New research provides insights into the treatment of Christianson syndrome (CS), an X-linked genetic disease characterized by reduced brain growth after birth, intellectual disability, epilepsy and difficulties with balance and speech.
“One of the major challenges in developing treatments for human brain disorders, like CS, is developing an experimental system for testing potential therapeutics on human neurons,” said study senior author Dr. Eric Morrow, an associate professor of molecular biology, neuroscience and psychiatry at Brown University. “In recent years, advanced stem cell therapies that use tissues from patients have provided powerful new approaches for engineering human neurons from the patients themselves, who may undergo the treatment in the future.”
For the study, published in Science Translational Medicine on Feb. 10, 2021, Morrow and his colleagues obtained blood samples from five CS patients and the patients’ unaffected brothers. They then reprogrammed these blood cells into stem cells, and these stem cells were converted into neurons in a petri dish. As a result, they obtained neurons that were representative of those from CS patients, and they used these neurons to test treatments.
Morrow—who directs the Center for Translational Neuroscience at the Carney Institute for Brain Scienceand the Brown Institute for Translational Science—said the team also used a new gene-editing approach that employs CRISPR-Cas9 technologies to correct patient mutations back to a healthy gene sequence.
CS is caused by a mutation in a gene encoding for NHE6, a protein that helps regulate acid levels within cell structures called endosomes. Past research suggests that the loss of NHE6 causes endosomes to become overly acidic, which disrupts the abilities of developing neurons to branch out and form connections in the growing brain.
Loss of this important protein can arise from a variety of gene mutations in patients. The majority of CS mutations are called nonsense mutations, which prevent NHE6 from being produced at all; four of the five CS patients involved in this study exhibited this class of mutation. However, some CS patients exhibit missense mutations. Individuals with missense mutations still have some NHE6, but it is produced in smaller amounts, and the protein fails to function as it should.
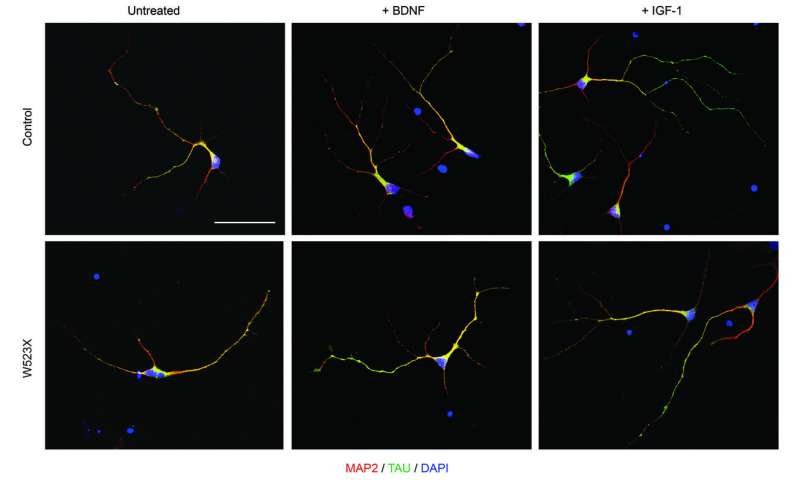
The research team tested two main forms of treatment on the stem-cell-derived neurons: first, gene transfer, which involves adding a healthy NHE6 gene into the cell; and second, administration of trophic factors, which are substances that promote neuron growth and encourage neurons to develop connections with other neurons. The researchers found that the neurons’ response to treatment depended on the class of mutation present.
The gene transfer studies, which may represent the first steps toward developing gene therapy, were successful in neurons with nonsense mutations. After the researchers inserted a functional NHE6 gene into nonsense-mutation CS neurons, the neurons branched out properly. In neurons with missense mutations, however, gene transfer failed completely. Further tests suggested that the abnormal NHE6 produced as a result of missense mutations may interfere with normal NHE6, thereby rendering gene transfer therapy ineffective in patient cells with these mutations.
In contrast, administration of trophic factors, such as brain-derived neurotrophic factor (BDNF) and insulin-like growth factor-1 (IGF-1), successfully promoted proper branching in all the CS neurons studied, regardless of mutation type.
While these initial results are encouraging, Morrow hopes that future studies will examine these treatments in animal models.
Source: Read Full Article